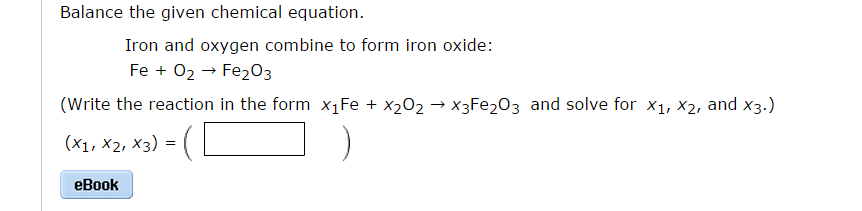
That is why we canceled the Na⁺ and Cl⁻ ions in the net ionic equation ( eq 3). The net ionic equation of reaction (2) is the following:Īll repeated ions in reactants and products do not participate in the net ionic equation. For example, C6H5C2H5 + O2 C6H5OH + CO2 + H2O will not be balanced, but XC2H5 + O2 XOH + CO2 + H2O will. Replace immutable groups in compounds to avoid ambiguity. Since the Al(OH)₃ precipitates, we cannot write it as an ion because it does not dissociate in the aqueous solution. Net Ionic Equation Calculator Instructions Use uppercase for the first character in the element and lowercase for the second character. To write the NET ionic equation, we need to write the total ionic equation:Īl³⁺(aq) + 3Cl⁻(aq) + 3Na⁺(aq) + 3OH⁻(aq) → Al(OH)₃(s) + 3Na⁺(aq) + 3Cl⁻(aq) (2) In this reaction, the ions Cl⁻ and OH⁻ are exchanged between the ions Al⁺³ and Na⁺. The reaction between AlCl₃ and NaOH is an exchange reaction:ĪlCl₃(aq) + 3NaOH(aq) → Al(OH)₃(s) + 3NaCl(aq) (1) Let’s demonstrate this by writing the ionic equation for the neutralisation of hydrochloric acid with sodium hydroxide.The balanced NET ionic equation for the reaction when AlCl₃ and NaOH are mixed in an aqueous solution is: Step 3: That’s it Now your window will display the Final Output of your Input. Finally, eliminate spectator ions and write the net ionic equation. Then write the ionic equation, showing all aqueous substances as ions. Step 2: For output, press the Submit or Solve button. Write and balance the molecular equation first, making sure that all formulas are correct. Step 1: In the input field, enter the required values or functions. Follow the below steps to get output of Ionic Equation Calculator. Therefore, the net ionic equation will show the actual chemical change, without the spectator ions. Steps to use Ionic Equation Calculator:. These are the spectator ions, which we cancel out. 
This is when you realise that some ions do not react: they remain dissociated in solution. Then, we rewrite the soluble ionic compounds as their dissociated ions. We can write ionic equations by starting from their chemical equations. Translating chemical equations into ionic equations (sodium, potassium, ammonium, or nitrate-containing) compounds are always soluble. Writing ionic equation lessons examples and solutions write a balanced net to show why the chegg com equations calculator tessshlo how total easy you 10 steps with pictures. If the table above feels overwhelming, just remember that S.P.A.N. digital marketing course free with certificate. Nevertheless, they are still useful in helping us to predict the solubility of common ionic compounds under normal circumstances. Obviously, even if sodium chloride is soluble, we cannot dissolve 1000 kg of it in 1 small drop of water. 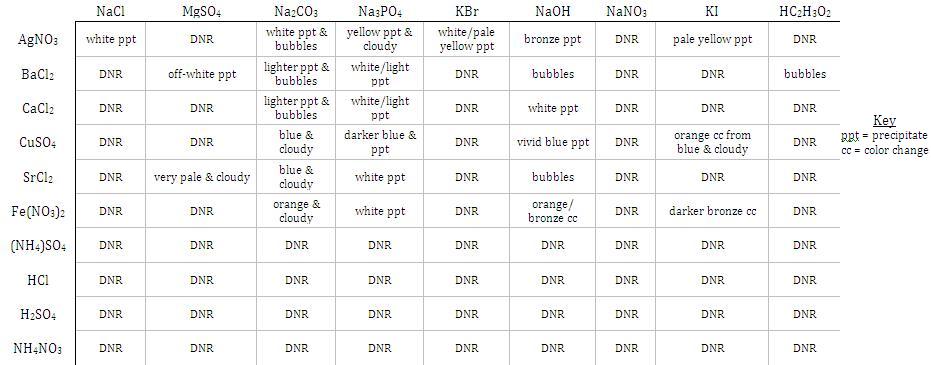
The actual solubility depends on temperature and the relative volume of water used. *The solubility rule for halides only apply to chlorides, bromides and iodides, but not fluorides. Using a Graphing Calculator Simplify the result to get the lowest, whole integer values. This means that we have to know what is soluble, and what is not. 
In ionic equations, we express dissolved ionic compounds as their dissociated ions, like Na + (aq) and Cl – (aq). The basics of writing ionic equations: knowing your solubility table This means that their ions are not dissociated. On the other hand, insoluble ionic compounds do not dissolve in water. In ionic equations, we will rewrite NaCl (aq) as Na + (aq) and Cl – (aq) separately, to emphasise that the ions are dissociated.ĭissociation is the separation of ions when a soluble ionic compound dissolves in water. The balanced NET ionic equation for the reaction when AlCl and NaOH are mixed in an aqueous solution is: Al³ (aq) + 3OH (aq) Al (OH) (s) The reaction between AlCl and NaOH is an exchange reaction: AlCl (aq) + 3NaOH (aq) Al (OH) (s) + 3NaCl (aq) (1) In this reaction, the ions Cl and OH are exchanged between the ions Al³ and Na. For example, we say that when sodium chloride dissolves in water, the sodium ions and chloride ions dissociate. The cheem word that we use for separate is dissociate. The separated ions are mobile, mixing around with the water molecules. When we dissolve them in water, the water molecules have a special ability to overcome the electrostatic forces of attraction, separating the ions. Yet, for soluble ionic compounds, their toughness is not without weakness. We usually describe these forces as “strong”. In the solid state, the ions of an ionic compound are held closely together by ionic bond. Ions of sodium chloride dissociate in water
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
